Leading the way to innovation
For over 40 years, MiniMed has committed to transforming diabetes care.
2016 - present
2026
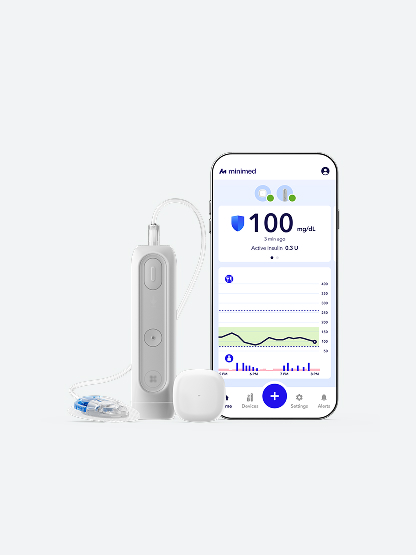
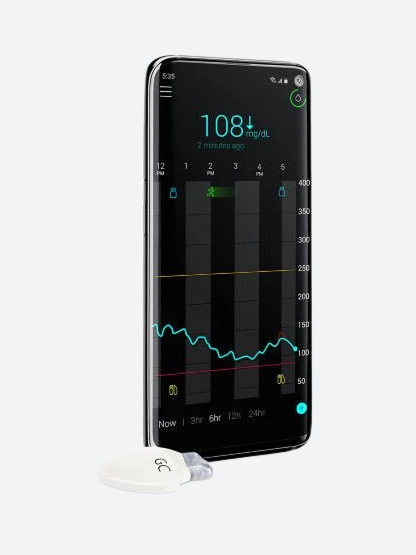
The MiniMed FlexTM insulin pump system
The MiniMed FlexTM system combines a screenless insulin pump with SmartGuardTM technology, a CGM, and the MiniMedTM app to support daily diabetes management with connected digital technology.
2025
Instinct sensor, made by Abbott
Exclusively for MiniMed™ systems, the Instinct sensor is a thin,1 discreet2 sensor designed for wear up to 15 days.
2025
Our first small all-in-one disposable sensor
Simplera Sync™ is an all-in-one disposable sensor designed to work with the MiniMed™ 780G system and SmartGuard™ technology.
2023
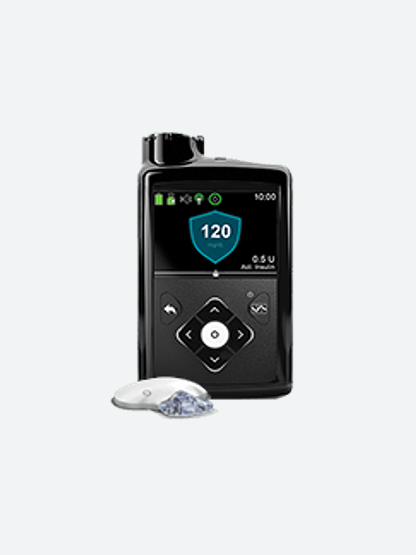
Meal DetectionTM technology
First insulin pump system with Meal DetectionTM technology‡ that delivers correction§ doses for miscalculated carb counts or occasionally missed meal doses.◊
2022
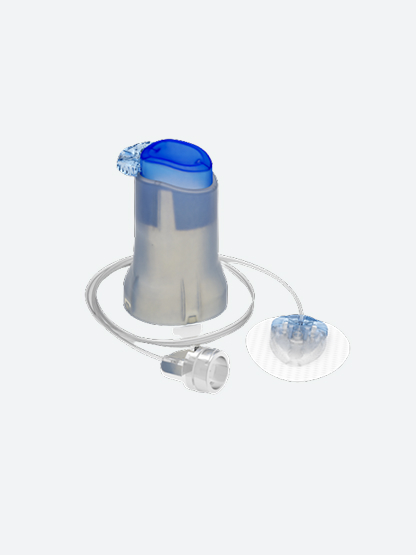
Medtronic ExtendedTM infusion set
First infusion set designed to be worn up to 7 days
2020
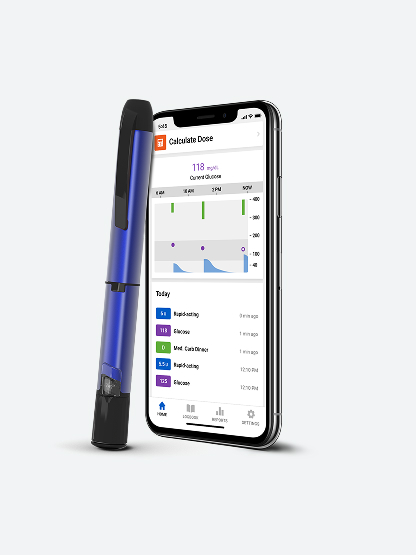
Smart insulin pen with CGM
First-ever real-time CGM and smart¶ insulin pen (real-time insulin injection insights)
2020
Insulin pump for kids
First automated insulin pump system approved for ages 2 and up
2018
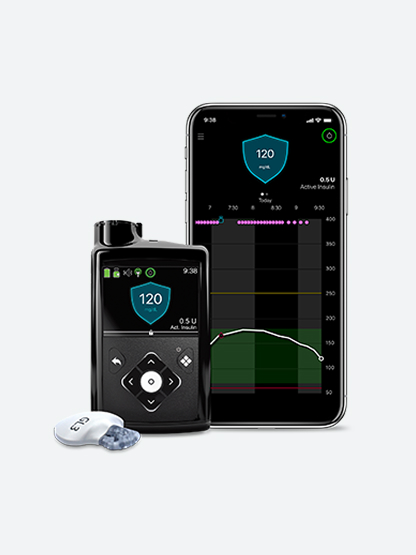
Continuous glucose monitor
First stand-alone smart CGM with predictive alerts for highs and lows#
2017
Hybrid closed-loop insulin pump
World’s first insulin pump system to automatically adjust background insulin
2016
Smart insulin pen
InPen™ smart insulin pen launched to improve diabetes management
Early 2000s - 2015
2015
Predictive algorithm∆
Breakthrough technology helps reduce lows with a predictive insulin suspend algorithm
2009
Technology to help reduce severe lows
First-ever insulin pump to automatically suspend insulin at a pre-set glucose target
2006
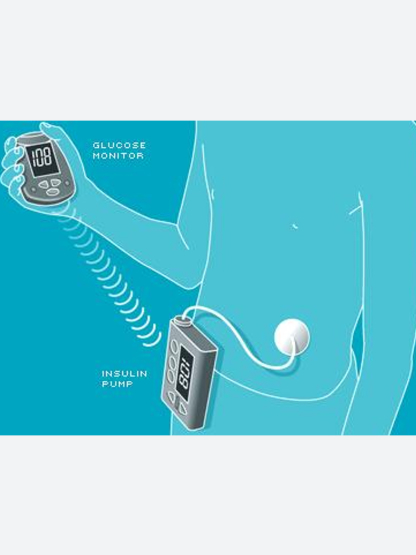
Insulin pump and CGM system
World’s first system to integrate insulin pump and CGM
2004
Guardian™ CGM system
The FDA approves the Guardian™ CGM system designed to notify users of potentially dangerous highs and lows by sounding an alarm.
2004
CareLinkTM delivers personalized reports
The CareLink™ Therapy Management system introduced a differentiated way to identify trends for therapy optimization. Reports are heavily leveraged to this day.
2003
World's first pump and CGM system
MiniMed™ Paradigm™ 512 pump and Paradigm Link® BG monitor becomes the world’s first system that allowed users to connect their CGM to their pump to deliver data live.
1990s
1999
First ever CGM system
FDA approval of first-ever CGM system — launching a new era in diabetes care.
1999
The MiniMed™ 508
Introduced the ability to administer or suspend insulin delivery remotely.
1998
MiniMed becomes Medtronic Diabetes
Medtronic acquires MiniMed Inc.
1998
The MiniMed™ 507
This launch marked a turning point in diabetes treatment and pump adoption, solidifying MiniMed as the leader in insulin pump therapy.
1992
The MiniMed™ 506
Major hardware redesign with advanced features such as meal bolus memory and daily insulin totals.
1980s
In the early 1980s, diabetes management was a daily struggle of multiple daily injections, unpredictable blood sugar levels, and constant vigilance. This was when Alfred E. Mann, a medical device pioneer and biotech entrepreneur, founded MiniMed with the introduction of the MiniMed™ 502 insulin pump. MiniMed’s early insulin pumps offered continuous subcutaneous insulin infusion (CSII), enabling improved blood sugar control. These pumps were smaller and more wearable compared to earlier, bulky designs. These early developments provided patients with an additional category of therapy options, enabling more freedom and flexibility in diabetes management. These pumps would lay the foundation for decades of new technology generations, through today’s more sophisticated AID systems.
Footnotes
‡ Taking a bolus 15 – 20 min before a meal helps to keep blood sugar levels under control after eating.
§ Refers to auto correct, which provides bolus assistance. Can deliver all correction doses automatically without user interaction, feature can be turned on and off.
◊ Refers to SmartGuard™ technology. Some user interaction required. Individual results may vary.
¶ Smart insulin pens connect to a mobile app to provide dosing calculations, reminders and CGM system integration.
# Refers to auto correct, which provides bolus assistance. Can deliver all correction doses automatically without user interaction, feature can be turned on and off.
∆ Only available outside of the USA.
Reference
1. Among patient-applied sensors.
2. Data on file, Abbott Diabetes Care, Inc.
Important Safety Information: InPen system
The InPen system consists of a reusable insulin pen and mobile app and is for people living with diabetes. It can be used to deliver insulin, help calculate insulin doses, and estimate carbohydrates for meals. Those under the age of 7 should only use the device with an adult’s supervision. A healthcare provider must prescribe InPen, provide dosage settings, and discuss all potential benefits and risks. Using the device with incorrect therapy settings may lead to severe highs and lows. The system should not be used by those unable to test blood glucose levels or the visually impaired. For additional safety information and user guides, see https://bit.ly/MiniMedRisks.
Important Safety Information: Extended Infusion Set
The Extended Infusion Set is indicated for up to 7 days of wear for the subcutaneous infusion of insulin from an infusion pump. It is NOT indicated for intravenous (IV) infusion or the infusion of blood or blood products. Inaccurate medication delivery, infection and/or site irritation may result from improper insertion and maintenance of the infusion site. Before insertion, clean the insertion site with isopropyl alcohol. Remove the needle guard before inserting the infusion set. If using this infusion set for the first time, do the first set-up in the presence of your healthcare professional. Do not leave air in the infusion set. Prime completely. Check frequently to make sure the soft cannula remains firmly in place as you may not feel pain if it pulls out. The soft cannula must always be completely inserted to receive the full amount of medication. If the infusion site becomes inflamed, replace the set, and use a new site until the first site has healed. Replace the infusion set if the tape becomes loose, or if the soft cannula becomes fully or partially dislodged from the skin. Regularly replace the infusion set as indicated in the instructions for use, or per the insulin labeling, whichever duration is shorter. For additional safety information and user guides, see https://bit.ly/MiniMedRisks.
Important Safety Information: MiniMedTM 780G system with SmartGuardTM technology with Instinct sensor, Simplera SyncTM sensor, and GuardianTM 4 sensor
The MiniMedTM 780G system is intended for the continuous delivery of basal insulin at selectable rates and the administration of insulin boluses at selectable rates for the management of type 1 diabetes mellitus in persons 7 years of age and older, and of type 2 diabetes mellitus in persons 18 years of age and older requiring insulin. The system is also intended to continuously monitor glucose vales in the fluid under the skin.
The MiniMedTM 780G System includes SmartGuardTM technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values. The system is intended for use with connected sensors, including the Simplera SyncTM and GuardianTM 4 sensors and integrated continuous glucose monitors, including the Instinct sensor, each of which has different wear-time, form factor, insertion site, and other distinguishing characteristics that relate to sensor performance. Consult the appropriate sensor user guide when using the system. Discuss treatment decisions with your HCP.
WARNING: Do not use the SmartGuardTM feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuardTM feature.
WARNING: Do not use MiniMedTM 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of MiniMedTM 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuardTM feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a blood glucose (BG) meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMedTM 780G system has not been studied in pregnant women or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including user guides and important safety information such as indications, contraindications, warnings and precautions associated with system and its components, see https://bit.ly/MiniMedRisks.
©2026 MiniMed. MiniMed and MiniMed logo are trademarks of Medtronic MiniMed, Inc. The sensor shape and appearance, Abbott, and “a” logo are marks and/or designs of the Abbott group of companies in various territories and used under license. Sensor image ©2026 Abbott. TM*Third–party brands are trademarks of their respective owners.
DreaMed Diabetes is a trademark of DreaMed Diabetes, Ltd. The MiniMedTM 780G system algorithm includes technology developed by DreaMed Diabetes.
.png)