Try the MiniMedTM 780G system at no cost†
Experience the difference of MiniMed automation and a 15-day sensor with our pump evaluation program.
How the program works
- Fill out the online form below.
- We’ll verify eligibility.
- We'll ship you the pump system to try for 30 days.
What you’ll get
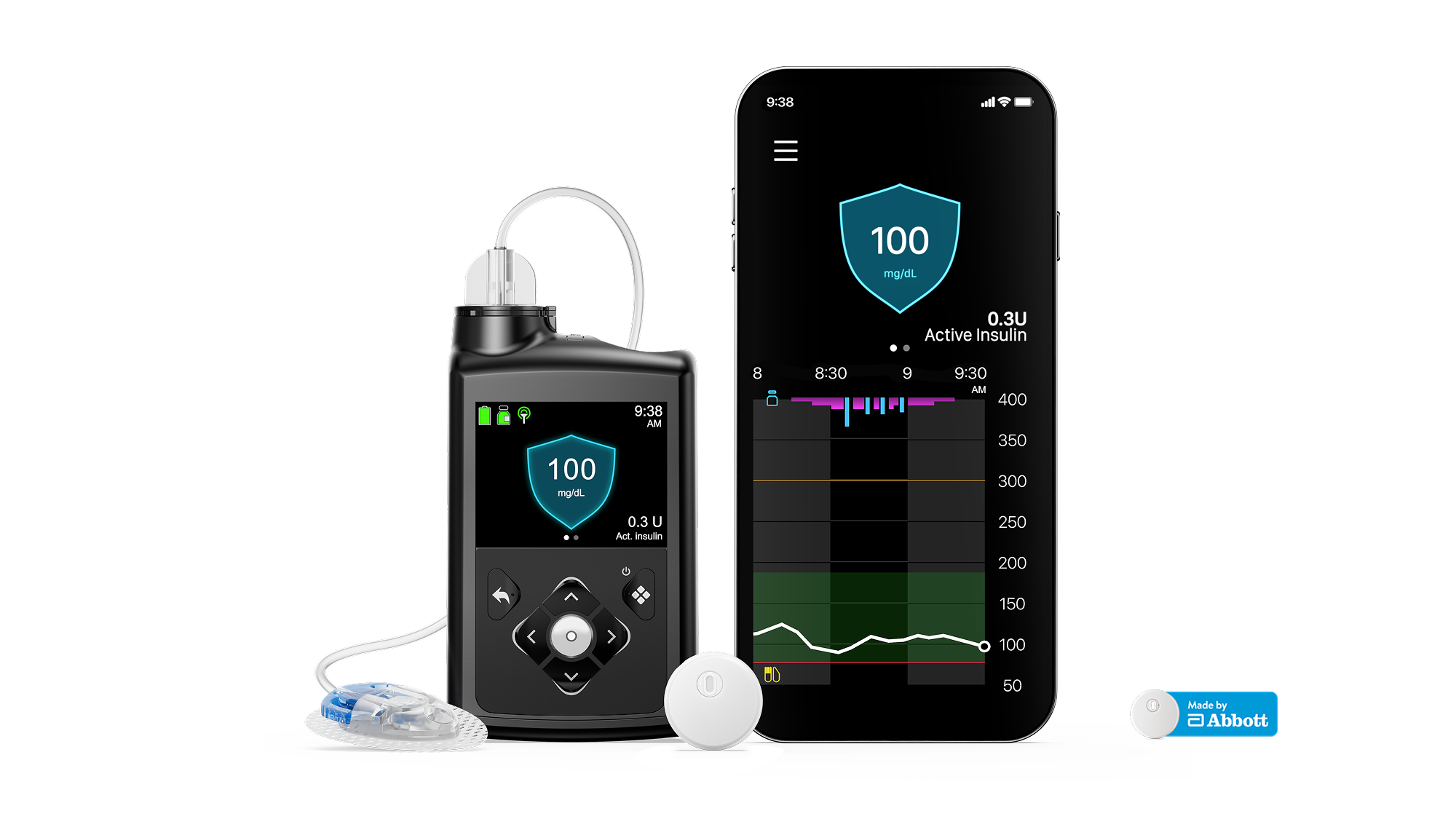
- 1 MiniMedTM insulin pump
- 1 month supply of the 15-day Instinct sensor, made by Abbott.
- 1 month supply of the 7-day Extended infusion set and reservoirs.
Options to choose another sensor or infusion set if you would like.
Switching from insulin injections to a pump can be easy

“I used to worry that wearing an insulin pump would make me feel constantly connected to my diabetes, but ironically, I now feel less connected to it than ever before. The pump has become so seamless in my life that I barely notice it, and this newfound ease has brought me a confidence I never expected.”
— Mattie, MiniMed Champion and long-time former insulin pen user.
Frequently asked questions
Am I eligible for this evaluation program?
If you have diabetes and are using commercial insurance, you may be eligible for this program. Terms and conditions apply.
What will I need to complete to participate in this program?
We will need some information about you to check eligibility, a signed agreement form, and a prescription from your healthcare professional.
If I’m on an older MiniMed pump, can I participate?
This program is for existing and new customers that have not tried the MiniMed 780G system.
Footnotes
† Terms and conditions
‡ Taking a bolus 15 – 20 minutes before a meal helps to keep blood sugar levels under control after eating.
Program Terms and Conditions:
- Eligibility is limited to customers new to the MiniMed™ 780G System.
- A prescription is required.
- Participation is excluded for residents of Massachusetts, Michigan, Rhode Island and Minnesota.
- Participation is excluded for individuals enrolled in any federal, state or government-funded healthcare programs (such as Medicare, Medicaid, Tricare, etc.), Kaiser Permanente programs, or Excellus health programs.
- Evaluation items must be returned to Medtronic Diabetes within 90 days after completion of the evaluation period and cannot be sold or submitted for third[1]party reimbursement.
- Term and conditions are subject to change. Additional terms and conditions apply, see Patient Evaluation Program Patient Agreement for details.
Important Safety Information: MiniMedTM 780G system with SmartGuardTM technology with Instinct sensor, Simplera SyncTM sensor, and GuardianTM 4 sensor
The MiniMedTM 780G system is intended for the continuous delivery of basal insulin at selectable rates and the administration of insulin boluses at selectable rates for the management of type 1 diabetes mellitus in persons 7 years of age and older, and of type 2 diabetes mellitus in persons 18 years of age and older requiring insulin. The system is also intended to continuously monitor glucose vales in the fluid under the skin.
The MiniMedTM 780G System includes SmartGuardTM technology, which can be programmed to automatically adjust insulin delivery based on the continuous glucose monitoring (CGM) sensor glucose values and can suspend delivery of insulin when the sensor glucose (SG) value falls below or is predicted to fall below predefined threshold values. The system is intended for use with connected sensors, including the Simplera SyncTM and GuardianTM 4 sensors and integrated continuous glucose monitors, including the Instinct sensor, each of which has different wear-time, form factor, insertion site, and other distinguishing characteristics that relate to sensor performance. Consult the appropriate sensor user guide when using the system. Discuss treatment decisions with your HCP.
WARNING: Do not use the SmartGuardTM feature for people who require less than 8 units or more than 250 units of total daily insulin per day. A total daily dose of at least 8 units, but no more than 250 units, is required to operate in the SmartGuardTM feature.
WARNING: Do not use MiniMedTM 780G system until appropriate training has been received from a healthcare professional. Training is essential to ensure the safe use of MiniMedTM 780G system.
WARNING: Do not use SG values to make treatment decisions, including delivering a bolus, while the pump is in Manual Mode. When the SmartGuardTM feature is active and you are no longer in Manual Mode, the pump uses an SG value, when available, to calculate a bolus amount. However, if your symptoms do not match the SG value, use a blood glucose (BG) meter to confirm the SG value. Failure to confirm glucose levels when your symptoms do not match the SG value can result in the infusion of too much or too little insulin, which may cause hypoglycemia or hyperglycemia.
Pump therapy is not recommended for people whose vision or hearing does not allow for the recognition of pump signals, alerts, or alarms. The safety of the MiniMedTM 780G system has not been studied in pregnant women or in persons using other anti-hyperglycemic therapies that do not include insulin. For complete details of the system, including user guides and important safety information such as indications, contraindications, warnings and precautions associated with system and its components, see https://bit.ly/MiniMedRisks.
©2026 MiniMed. MiniMed and MiniMed logo are trademarks of Medtronic MiniMed, Inc. The sensor shape and appearance, Abbott, and “a” logo are marks and/or designs of the Abbott group of companies in various territories and used under license. Sensor image ©2026 Abbott. TM*Third–party brands are trademarks of their respective owners.
DreaMed Diabetes is a trademark of DreaMed Diabetes, Ltd. The MiniMedTM 780G system algorithm includes technology developed by DreaMed Diabetes.
.png)
